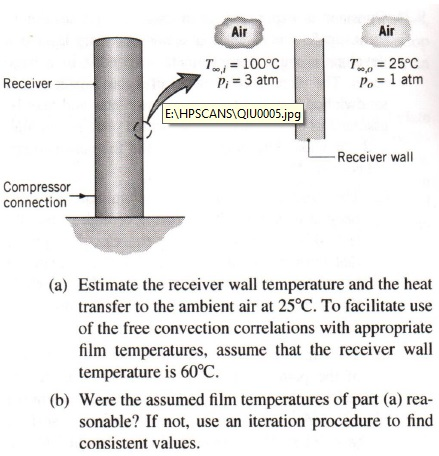
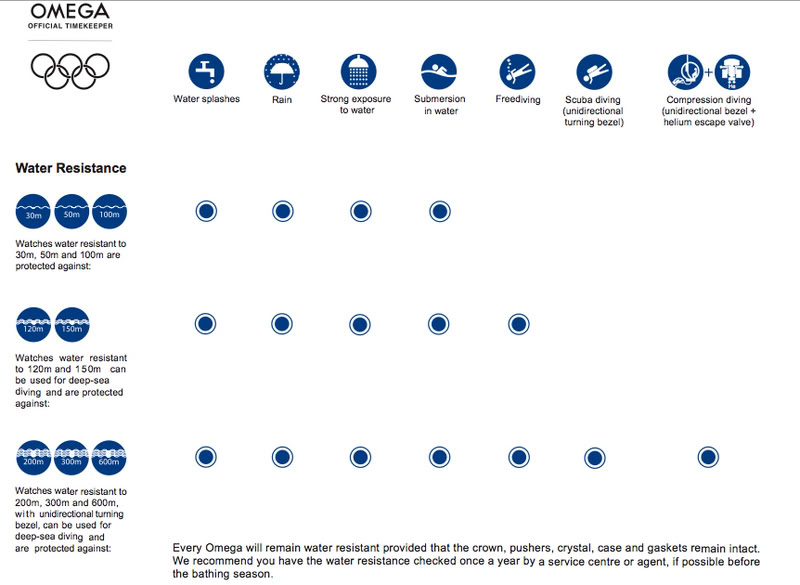
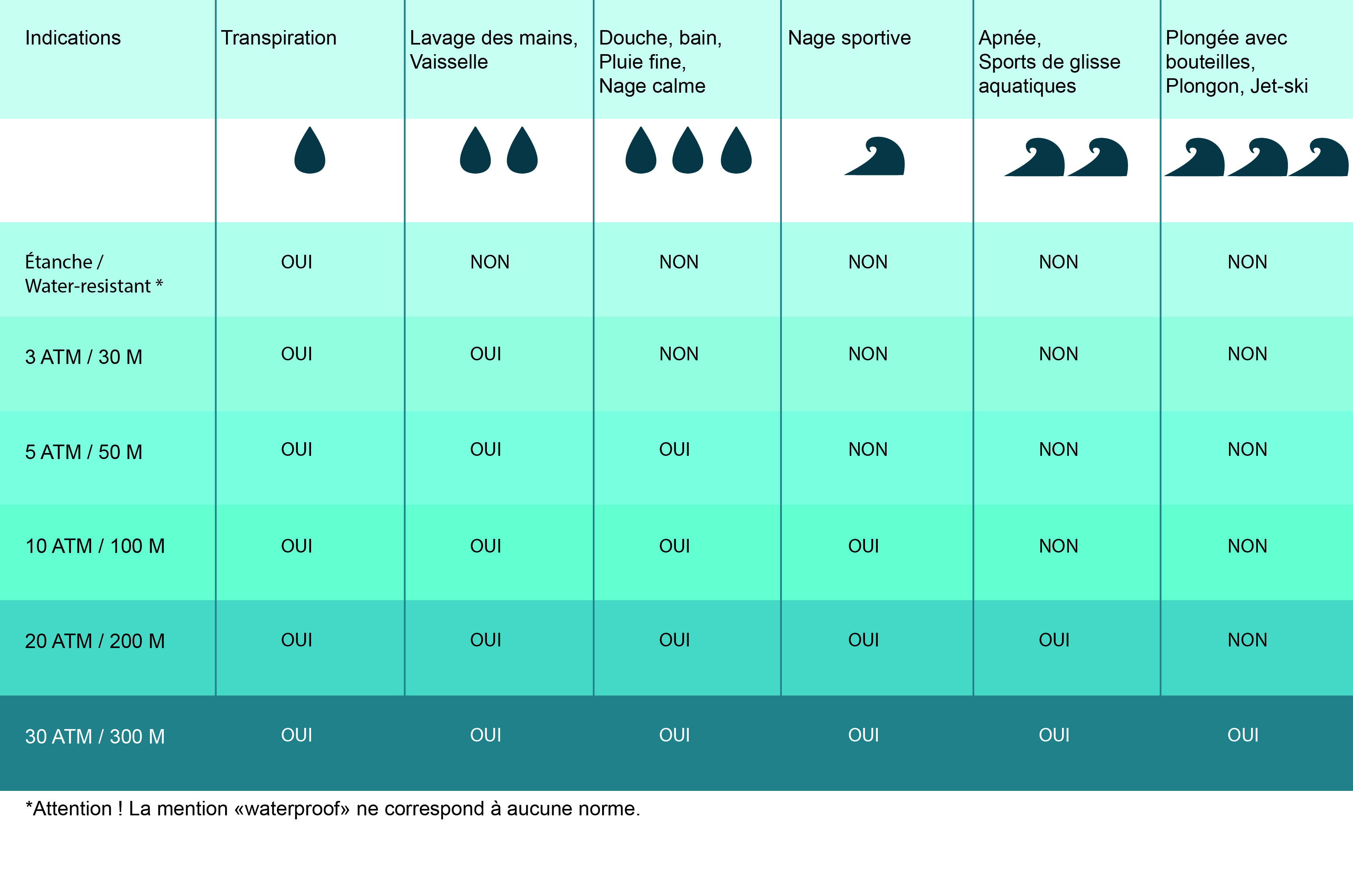
A hole is made at the bottom of a tank filled with water (density = 10^3 kg/ m^3) . If the total pressure at the bottom of the tank is 3 atm (

EXRIZU F3 Montre Smart Watch IP67, Étanche 3ATM Soutien Bluetooth, Lumineux ,Altimètre, Podomètre - Meshago Niger

Welcome to Chem Zipper.com......: A balloon of diameter 20 m weighs 100 kg. Calculate its paylod if it is filled with helium at 1.0 atm and 27 ∘ C. Density of air

Self-contained underwater breathing apparatus diving and asthma - Annals of Allergy, Asthma & Immunology

The Henry's law constant for oxygen gas in water at `25^(@)C` is `1.3xx10^(- 3)` M `atm^(-1)`. - YouTube
![A total pressure of \\[3\\] atm when applied on the surface of solution of concentration \\[0.11\\] M prevents flow of solvent across the semipermeable membrane from the other side of solution of A total pressure of \\[3\\] atm when applied on the surface of solution of concentration \\[0.11\\] M prevents flow of solvent across the semipermeable membrane from the other side of solution of](https://www.vedantu.com/question-sets/e8f3c919-2a2b-4f85-a689-9d36f461c6642200503482266048266.png)